Beyond the Scale: Diabolic Ways Obesity Affects Your Body's Core Systems
Most of us think of obesity in terms of weight, body mass index (BMI), and its well-known links to chronic conditions like type 2 diabetes and heart disease. But this view only scratches the surface. The most profound impacts of obesity often happen silently, deep within the body's hormonal, cellular, and even genetic systems.
This article reveals five of these lesser-known, impactful ways that excess body fat, particularly when stored in specific locations, can fundamentally alter our health from the inside out.
1. It’s Not the Pounds, It's the Placement: The Danger of "Hidden" Fat
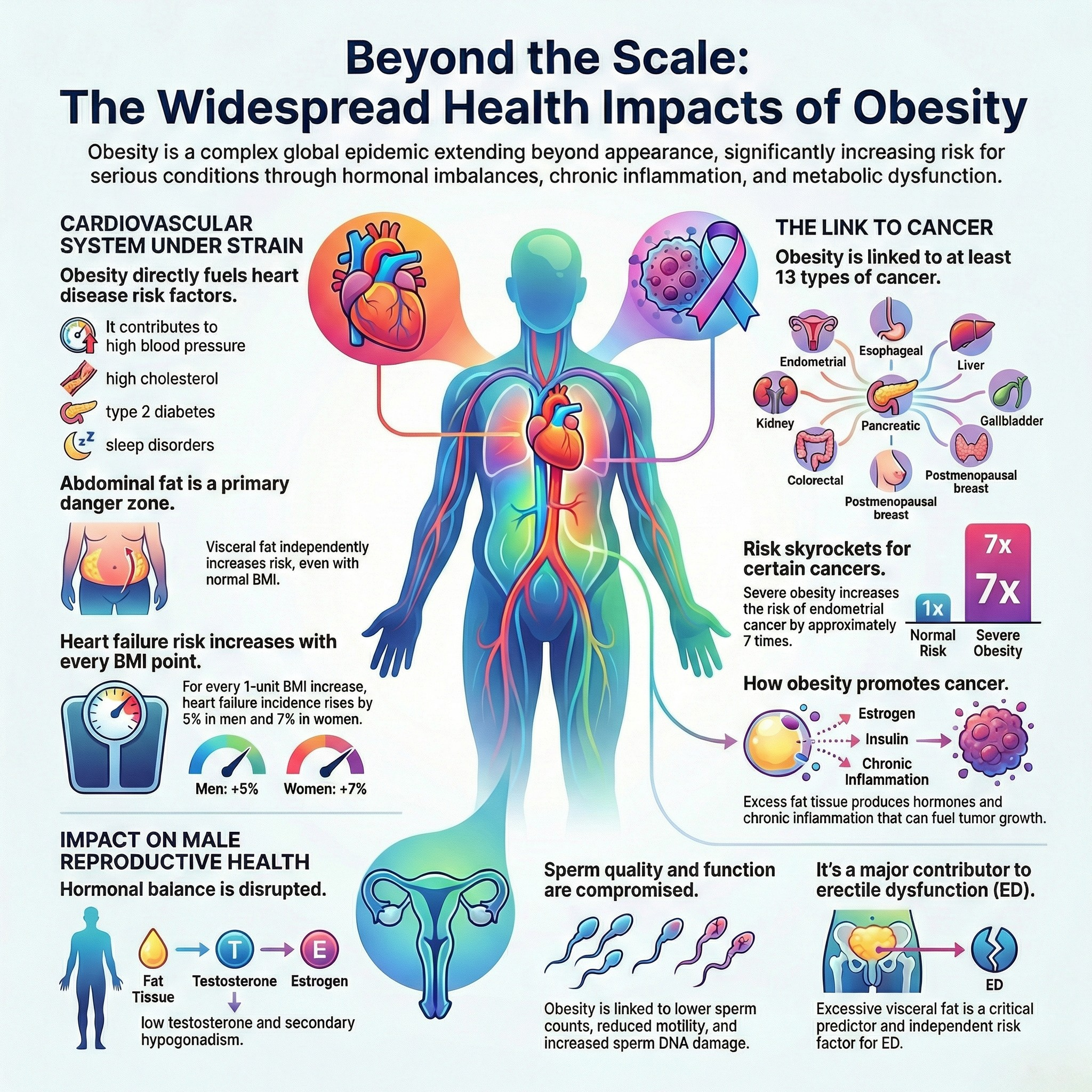
While Body Mass Index (BMI) is a common measure, it doesn't tell the whole story about health risks. The location of your body fat is far more critical than the total number on a scale. The real danger lies in abdominal obesity, specifically the accumulation of visceral adipose tissue (VAT)—the "hidden" fat surrounding your internal organs.
Having an "apple-shaped" body with a large waist circumference is a key indicator of high VAT and a core component of diagnosing metabolic syndrome. According to the American Heart Association, a high waist circumference, even in individuals with a normal weight, may unmask higher cardiovascular disease risk. This condition, sometimes called "normal-weight obesity," highlights why a tape measure can provide critical information that BMI alone misses. Other forms of "ectopic fat" (fat stored in the wrong places), such as deposits around the heart (pericardial/epicardial fat) and in the liver, are also directly linked to poor cardiovascular outcomes. This crucial distinction shifts the focus from simply losing weight to the more important goal of improving body composition and reducing central fat for true health benefits.
2. A Hormonal Hijacking: How Obesity Rewires Male Biology
Far from being inert storage, fat tissue is a massive and metabolically active endocrine organ—and in men with obesity, it can function like a rogue hormonal factory. Adipose (fat) tissue contains an enzyme called aromatase, which irreversibly converts testosterone into estradiol, a form of estrogen.
Because this enzyme resides in the fat tissue itself, a greater volume of fat literally creates more factories for this conversion. In men with obesity, the resulting elevated aromatase activity leads to two key changes: testosterone levels decrease while estrogen levels rise. This hormonal imbalance is not a minor fluctuation; it has direct and serious consequences, including:
Secondary Hypogonadism: A condition where the testes produce insufficient testosterone.
Impaired Male Fertility: This can manifest as poor sperm quality and erectile dysfunction.
Metabolic Disruption: The hormonal shift is linked to insulin resistance and disrupts the body's ability to maintain stable blood sugar levels (glucose homeostasis).
This illustrates how obesity can fundamentally alter a man's hormonal identity, with profound consequences for both his metabolic health and reproductive capability.
3. Creating a Cancer-Friendly Environment
The link between obesity and cancer is not just a statistical correlation; obesity actively creates conditions in the body that help cancer develop and grow. Researchers have identified several clear biological mechanisms that explain this connection, with consistent evidence now linking higher amounts of body fat to an increased risk of at least 13 different types of cancer.
Here are three key ways obesity fosters a cancer-friendly internal environment:
First, fat tissue functions like an endocrine organ, producing excess amounts of estrogen. High levels of this hormone are known to increase the risk of developing breast, endometrial, and ovarian cancers.
At the same time, people with obesity often develop insulin resistance, which leads to higher circulating levels of insulin and insulin-like growth factor-1 (IGF-1). Both substances can act as a growth accelerator for cells, which unfortunately can't distinguish between healthy cells and cancerous ones, promoting tumor growth.
Finally, this environment is maintained by a state of chronic, low-grade inflammation. Fat cells in individuals with obesity can promote this persistent inflammation, which is a known driver of cancer because it can directly promote tumor growth.
4. An Unwanted Inheritance: Passing Health Risks to the Next Generation
Beyond our DNA, our bodies have a second layer of instructions known as epigenetics—changes that affect how genes work without altering the DNA sequence itself. One of the most startling recent discoveries is that a father can pass these instructions, and his health risks, on to his children.
Research shows that diet-induced obesity in a father can lead to epigenetic modifications in his sperm, including alterations to DNA methylation, the way DNA is packaged with histone proteins, and the composition of sperm RNA. These epigenetic changes can be inherited and may disrupt early embryonic growth, with studies showing paternal obesity can negatively influence embryos at various developmental stages. This may ultimately predispose the offspring to metabolic dysfunction later in life.
The implication is profound. This means a father's metabolic health at the time of conception is not just a personal matter; it may be writing a biological story for a child who hasn't even been conceived yet.
5. The Counter-intuitive "Obesity Paradox"
While obesity is a primary cause of cardiovascular disease (CVD), a strange and counter-intuitive phenomenon has been observed in people who already have an established condition like heart failure or coronary artery disease. This phenomenon is known as the "obesity paradox." In some studies of patients with existing CVD, those who are overweight or have class 1 obesity (BMI 30-34.9) sometimes show better short-term survival rates than their normal-weight counterparts.
However, it is crucial to interpret this finding with extreme caution. This is a complex statistical observation, not a recommendation to gain weight. Scientists have proposed several potential explanations, including:
Lead Time Bias: People with obesity may be diagnosed with CVD at an earlier age, giving them a head start on treatment compared to leaner individuals who are diagnosed later.
Frailty and Cachexia: In patients who are already sick, a low body weight can be a dangerous sign of severe illness, frailty, or "cardiac cachexia"—a condition of extreme muscle and fat wasting caused by advanced heart disease. The "normal-weight" group in these studies may be artificially skewed by these very frail individuals, making the overweight group appear healthier simply by comparison.
Ultimately, the long-term, well-established health risks of obesity far outweigh any observed short-term statistical paradox.
Conclusion: A Deeper Definition of Health
Our journey has taken us from the visible placement of fat on our bodies to the invisible, internal environments of our cells, the surprising complexities of disease statistics, and even into the health of future generations. These five examples reveal that obesity's true impact is systemic and metabolic, affecting everything from our hormonal balance and cellular function to the epigenetic legacy we pass on. This deeper understanding challenges us to look beyond the scale and focus on the internal processes that truly define our well-being.
Knowing that health is determined more by our metabolic function than the number on a scale, how might we redefine our approach to wellness and disease prevention?